2 Consider the following Reaction SiCl4 + 3Ch3MgCl > P (Compound with silicon) P + H2O >Q + HCl 2Q > H20 S Select the correct statement about compound S. 1) S

Oxidation of Liquid Silicon in Air Atmospheres Containing Water Vapor | Industrial & Engineering Chemistry Research
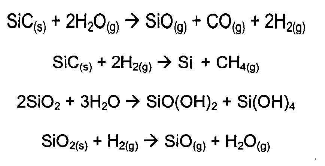
SiC and SiO2 may chemically react with H2O and H2 - graphite-graphite felt-carbon composite-manufacturer-CFCCARBON LTD

How to Balance Si+H2O= SiO2+ H2| Reaction Balance Si+H2O=SiO2+H2| Chemical equation Si+H2O=SiO2+H2 - YouTube
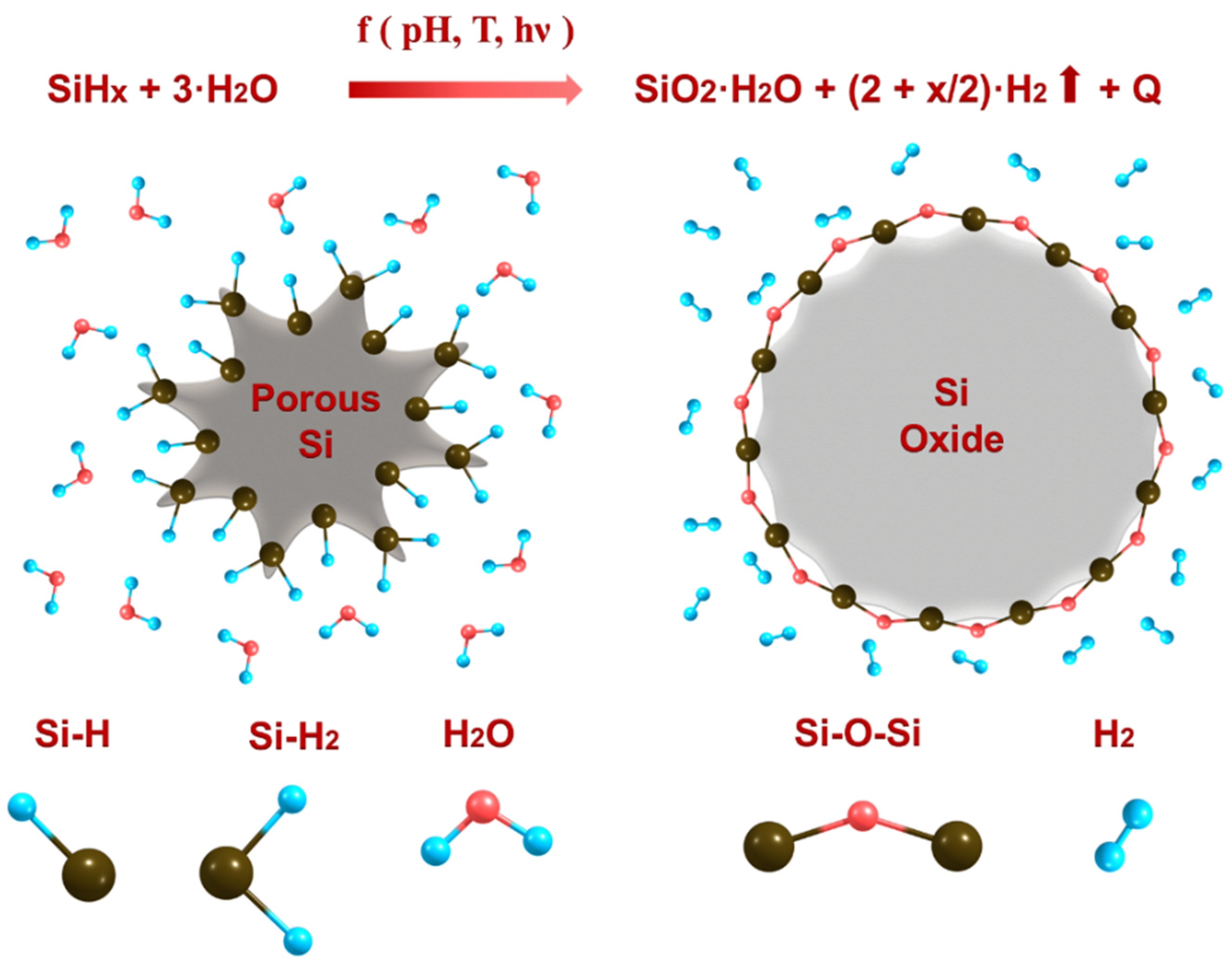
Nanomaterials | Free Full-Text | Kinetics of Hydrogen Generation from Oxidation of Hydrogenated Silicon Nanocrystals in Aqueous Solutions

SOLVED: 'Arrange the following in order of increasing melting point (lowest first): CO2, Si, H2O, and NaCl CO2, H2O, Si, NaCl H2O, CO2, NaCl, Si CO2, H2O, NaCl; Si NaCl,; Si, CO2,H2O
![PDF] Acid dissociation mechanisms of Si(OH)(4) and Al(H2O)(6)(3+) in aqueous solution | Semantic Scholar PDF] Acid dissociation mechanisms of Si(OH)(4) and Al(H2O)(6)(3+) in aqueous solution | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/ddadef5594a7bd51f0ffbb59d966191985b308d3/6-Table1-1.png)
PDF] Acid dissociation mechanisms of Si(OH)(4) and Al(H2O)(6)(3+) in aqueous solution | Semantic Scholar

Tunable Interfacial Electronic Pd–Si Interaction Boosts Catalysis via Accelerating O2 and H2O Activation | JACS Au







![SiCl4 [ ]H2O (A) [ ]Heat (B) [ Heat ]+Na2CO3 (C) . The compound (C) is: SiCl4 [ ]H2O (A) [ ]Heat (B) [ Heat ]+Na2CO3 (C) . The compound (C) is:](https://haygot.s3.amazonaws.com/questions/1693075_1728586_ans_27407beffa814a64a6cfde0548a6a066.jpeg)





